Untreated celiac disease complications arise when continued gluten ingestion triggers chronic intestinal inflammation. This damages the small intestine’s villi, leading to severe nutrient malabsorption, osteoporosis, and iron-deficiency anemia. Long-term risks include the development of additional autoimmune disorders, infertility, neurological damage (gluten ataxia), and a slightly increased risk of intestinal cancers like lymphoma.
Celiac disease is far more than a simple food intolerance; it is a serious, lifelong autoimmune condition where the ingestion of gluten leads to damage in the small intestine. For the estimated 1% of the global population affected, a strict gluten-free diet is the only effective treatment. However, when the condition remains undiagnosed or the diet is not strictly followed, the consequences extend well beyond gastrointestinal distress.
The long-term risks associated with non-compliance or delayed diagnosis are systemic, affecting nearly every organ system in the body. Understanding these risks is crucial for patient compliance and long-term health management.
What are the risks of Nutrient Malabsorption and Osteoporosis?
The primary mechanism of injury in celiac disease is villous atrophy—the flattening of the finger-like projections in the small intestine responsible for nutrient absorption. When these villi are damaged due to untreated celiac disease complications, the body loses its ability to absorb essential vitamins and minerals, regardless of how healthy the patient’s diet may be. This state of malabsorption is the precursor to several debilitating conditions.
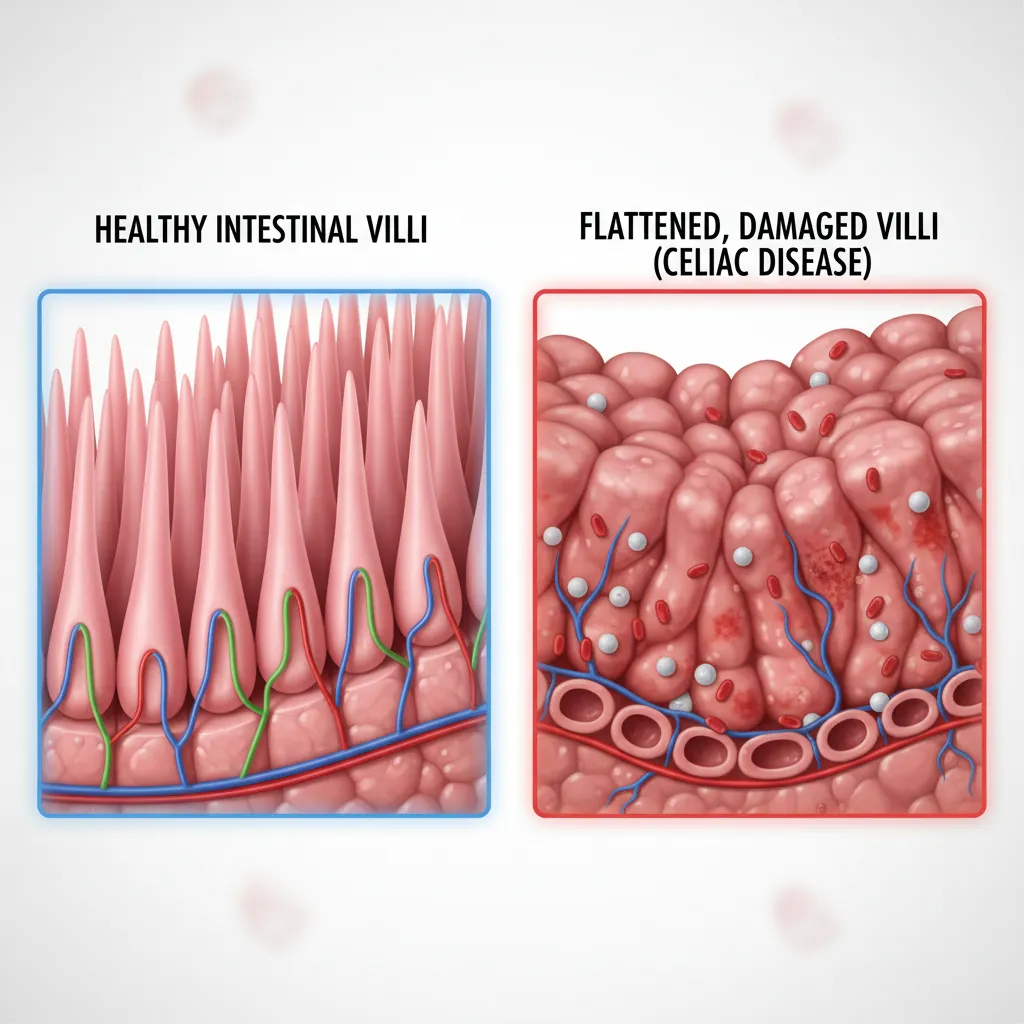
Iron Deficiency Anemia
One of the most common extra-intestinal manifestations of untreated celiac disease is iron deficiency anemia. Unlike typical anemia, which might be resolved with oral supplementation, anemia caused by celiac disease is often resistant to oral iron therapy. This is because the duodenum—the primary site of iron absorption—is often the first section of the intestine to be damaged by gluten exposure.
Patients often present with unexplained fatigue, weakness, and pallor long before digestive symptoms appear. If the underlying autoimmune reaction is not halted through a strict gluten-free diet, iron stores cannot be replenished, leading to chronic hypoxia and strain on the cardiovascular system.
Osteopenia and Osteoporosis
Bone health is critically compromised in untreated celiac disease due to the malabsorption of two key nutrients: calcium and Vitamin D. Furthermore, chronic inflammation leads to the release of pro-inflammatory cytokines, which can increase bone resorption (breakdown) over bone formation.
Research indicates that adults with undiagnosed or untreated celiac disease have significantly lower bone mineral density (BMD) than the general population. This progresses from osteopenia (lower than normal bone density) to full-blown osteoporosis, drastically increasing the risk of fractures. In children, this malnutrition results in rickets and failure to thrive, potentially permanently stunting growth.
Vitamin B12 and Folate Deficiency
Damage to the ileum can impair the absorption of Vitamin B12 and folate. Deficiencies in these vitamins can lead to macrocytic anemia and peripheral neuropathy. Symptoms may include tingling in the hands and feet, cognitive difficulties, and mood disturbances. Correcting these levels requires not only supplementation but also the healing of the intestinal lining to restore absorptive capacity.
How does untreated Celiac Disease trigger other Autoimmune Conditions?
Autoimmune disorders tend to cluster. When the immune system is already hyperactive and attacking the body’s own tissues (as in celiac disease), the likelihood of developing a second or third autoimmune condition increases significantly. The risk is directly correlated with the duration of gluten exposure; the longer a patient goes undiagnosed or untreated, the higher the probability of developing associated conditions.

Thyroid Disease (Hashimoto’s and Graves’)
Thyroid disorders are the most common autoimmune conditions associated with celiac disease. Hashimoto’s thyroiditis (hypothyroidism) and Graves’ disease (hyperthyroidism) occur more frequently in celiac patients than in the general population. The shared genetic susceptibility (HLA-DQ2 and HLA-DQ8 markers) plays a role, but the systemic inflammation caused by untreated celiac disease is a key trigger. Managing thyroid medication levels can be difficult if the gut is damaged, as the medication itself may not be absorbed consistently.
Type 1 Diabetes
There is a well-established link between Type 1 Diabetes (T1D) and celiac disease. It is estimated that 4-9% of patients with T1D also have celiac disease. For these patients, untreated celiac disease complications are particularly dangerous because malabsorption can lead to unpredictable blood glucose levels, increasing the risk of both hypoglycemia and diabetic ketoacidosis. Regular screening for celiac disease is now a standard of care for T1D patients.
Sjogren’s Syndrome and Liver Disease
Untreated celiac disease is also linked to Sjogren’s syndrome (characterized by dry eyes and mouth) and autoimmune hepatitis. Elevated liver enzymes are frequently seen in undiagnosed celiac patients. In most cases, these liver enzymes return to normal ranges after adopting a strict gluten-free diet, highlighting the liver’s sensitivity to the systemic inflammation caused by gluten.
What are the Neurological and Reproductive Risks?
While celiac disease is defined by intestinal damage, its impact on the nervous and reproductive systems is profound and often irreversible if left untreated for too long.
Gluten Ataxia and Neuropathy
Gluten ataxia is a condition where the immune system attacks the cerebellum, the part of the brain responsible for motor control. This results in loss of balance, coordination, and slurred speech. Unlike intestinal villi, which can regenerate, neural tissue has limited regenerative capacity. Therefore, early diagnosis is vital to prevent permanent neurological disability. Peripheral neuropathy (numbness and pain in extremities) is another common complication, often exacerbated by B12 deficiency.

Infertility and Pregnancy Complications
For women, untreated celiac disease is a known cause of unexplained infertility, recurrent miscarriages, and intrauterine growth restriction (IUGR). The mechanisms involve both nutrient deficiencies (such as folic acid and zinc) and the systemic inflammatory response which may interfere with implantation and placental function. Men with untreated celiac disease may also experience gonadal dysfunction and altered sperm quality. Adherence to a gluten-free diet has been shown to restore fertility in many cases.
What is Refractory Celiac Disease and Malignancy?
In rare cases, the body fails to heal despite a strict diet, or the damage leads to malignant transformation. These are the most severe untreated celiac disease complications.
Refractory Celiac Disease (RCD)
Refractory Celiac Disease occurs when symptoms and intestinal damage persist or recur despite strict adherence to a gluten-free diet for at least 12 months. RCD is categorized into Type I and Type II. Type I usually responds to steroids and immunosuppressants. Type II is more severe, characterized by the presence of abnormal T-cells in the intestine, and is considered a precursor to lymphoma.
Enteropathy-Associated T-Cell Lymphoma (EATL)
EATL is a rare but aggressive form of non-Hodgkin lymphoma affecting the small intestine. The risk of developing EATL is significantly higher in patients with untreated celiac disease compared to the general population. However, the risk drops dramatically—approaching that of the general population—after five years of strict adherence to a gluten-free diet. This underscores the life-saving importance of dietary compliance.
Annual Check-up Checklist and Follow-up Care
Managing celiac disease requires more than just avoiding bread. Regular medical follow-up is essential to monitor mucosal healing, detect nutritional deficiencies early, and screen for associated autoimmune conditions. A reactive approach is insufficient; proactive management is required.

The Essential Annual Checklist
Patients should schedule a dedicated celiac follow-up appointment with a gastroenterologist or a knowledgeable primary care physician at least once a year. The following protocols are recommended:
- Serology (Antibody Testing):
- Tissue Transglutaminase IgA (tTG-IgA): To monitor compliance and immune response. Levels should decrease and normalize on a gluten-free diet.
- Deamidated Gliadin Peptide (DGP): Useful if tTG is equivocal or in young children.
- Nutritional Panel:
- Complete Blood Count (CBC) to check for anemia.
- Ferritin (Iron stores).
- Vitamin B12 and Folate.
- Vitamin D (25-hydroxy).
- Calcium and Zinc levels.
- Thyroid Screening:
- TSH (Thyroid Stimulating Hormone) to screen for autoimmune thyroid disease.
- Bone Density Scanning:
- DEXA scan recommended for adults at diagnosis and repeated every 2-3 years if osteopenia/osteoporosis is present.
- Dietary Review:
- Consultation with a specialized dietitian to review potential sources of cross-contact and ensure nutritional adequacy.
Adhering to this follow-up schedule ensures that any complications are caught early. If antibody levels remain elevated, it suggests ongoing gluten exposure (often inadvertent), requiring a thorough review of the patient’s environment and diet. Healing the intestine is the only way to mitigate the severe long-term risks associated with this condition.
People Also Ask
Can celiac disease cause death if untreated?
Yes, while rare in the short term, untreated celiac disease can lead to life-threatening complications over time. These include severe malnutrition, organ failure, and a specific type of intestinal cancer called Enteropathy-Associated T-Cell Lymphoma (EATL). However, strict adherence to a gluten-free diet reduces mortality rates to near-normal levels.
How long does it take for celiac damage to heal?
Healing time varies by individual and age. In children, intestinal villi may heal within 3 to 6 months of starting a gluten-free diet. In adults, full mucosal healing often takes longer, ranging from 2 years to 5 years. Some adults may never achieve complete histological recovery but can still manage symptoms effectively.
Can you have mild celiac disease with no complications?
No, there is no such thing as “mild” celiac disease regarding internal damage. Even if a patient has no outward symptoms (asymptomatic or “silent” celiac), the autoimmune reaction still damages the intestine and prevents nutrient absorption, leading to the same long-term risks like osteoporosis and anemia as those with severe symptoms.
What are the neurological symptoms of untreated celiac disease?
Neurological symptoms can be severe and include gluten ataxia (loss of balance and coordination), peripheral neuropathy (tingling or numbness in hands and feet), severe migraines (
